Decay Statistics
Activity
Definition
Activity is the rate of radioactive decay measured as the number of disintegrations per unit of time. Mathematically, activity may be defined as in the below equation where ΔN/Δt is the change in number of radioactive atoms (N) per unit time.
Key Point: The unit of Curie, 3.7E10 dps, is approximately the activity of 1 gram of Radium-226.
Equation
![]()
Units
Activity is measured in Curie (Ci), becquerel (Bq), or disintegrations per second (dps).
![]()
![]()
Decay Constant (λ)
The decay constant (λ) is the fraction of atoms of a radionuclide which decay in a given time.
![]()
Given the decay constant and the number of initial atoms of a radionuclide, the number of atoms remaining may be calculated as in the below equation.
![]()
Half-Life (T1/2)
Half life is the time required for half of the atoms of a radioactive sample to decay.
![]()
Decay rate is often expressed in terms of half lives as it provides a quick way to determine remaining activity using the below equation where t is the time elapsed.
![]()
Effective Biological Half-Life
Biological half life is defined as the time required to remove half of a substance from the body. For the case of the radionuclides, the effective biological half-life is a function of both biological processes and radioactive decay.
![]()
| Source | Half-Life |
| Radium-226 | 1622 years |
| Iridium-192 | 73.8 days |
| Cobalt-60 | 5.3 years |
| Cesium-137 | 30 years |
| Gold-198 | 2.7 days |
| Iridium-192 | 73.83 days |
| Iodine-125 | 59.4 days |
| Palladium-103 | 16.99 days |
Mean Life (Tmean)
Mean life is the average lifetime for the decay of radionuclide atoms.
![]()
Mean life can be used to quickly compute the total number of decays over the lifetime of the sample, which is equal to the initial number of atoms in the sample, N0.
![]()
Mean life is also useful as it allows simple calculation of cumulative dose from a permanent implant given an initial dose rate.
![]()
Radioactive Equilibrium
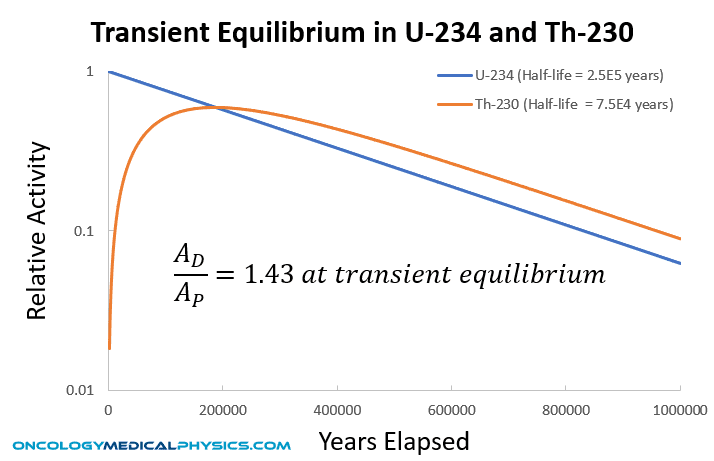
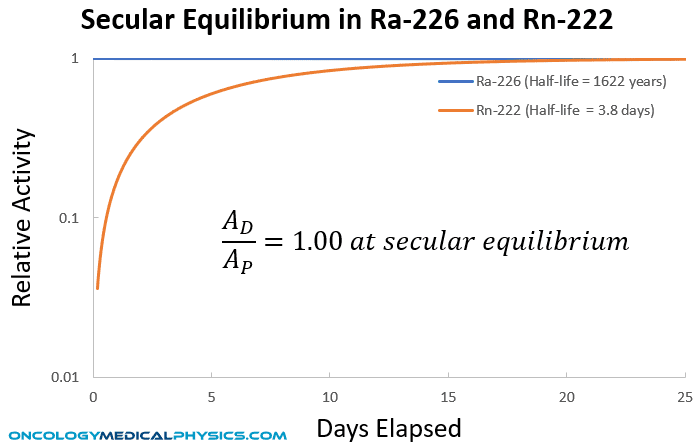
The product of decay for many radionuclides is another radioactive product with a distinct half life. In such radioactive decays, the original radionuclide is referred to as the parent and all subsequent radionuclide products are called daughters. If the half life of the parent is shorter than the daughters, no equilibrium will be reached between the parent and daughters. However, if the half life of the parent is longer than the daughters, an equilibrium in concentration of the parent and daughter will be established. Under conditions of equilibrium, the activity of the daughter nuclides will be governed by the disintegration rate of the parent.
Transient Equilibrium
[T1/2(parent) > T1/2(daughter)]
![]()
![]()
- TD is half-life of daughter
- TP is half-life of parent
- BR is branching ratio, the fraction of decays yielding the desired daughter nuclide
Once transient equilibrium has been achieved, the ratio of activity between the daughter and parent (AD/AP) is:
![]()
Maximum Daughter Activity
Maximum daughter activity is reached at time tmax.
![]()
Navigation
Not a Member?
Sign up today to get access to hundreds of ABR style practice questions.